Everything about calcium sulfate

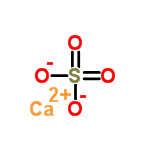
Calcium sulfate with the chemical formula CaSO4 is a mineral the hydrated form of which is called gypsum and plaster of Paris. It is a very useful compound in its hydrated and anhydrous forms. Calcium sulfate consists of one calcium atom, one sulfur atom and four oxygen atoms. It is an ionic compound that contains a cation (Ca+2) and a polyatomic anion (SO4-2). The valency of calcium ion is +2 whereas that of the polyatomic sulfate ion is -2. Consequently, CaSO4 is a neural compound.
The sulfate ion is a weak conjugate base of sulfuric acid (formerly called hydrogen sulfuric acid). Therefore, the sulfate ion does not decrease the pH of a solution.
General information about calcium sulfate
| Chemical name | Calcium sulfate |
| Other names | Gypsum, anhydrous calcium sulfate |
| English synonyms | Plaster of Paris, Drierite, gypsum |
| Molecular formula | CaSO4 |
| Molecular mass | 136.14 g/mol |
Hydrated forms of calcium sulfate
Calcium sulfate hemihydrate or plaster of Paris (CaSO4.0.5H2O)
Calcium sulfate dihydrate (CaSO4.2H2O)
Anhydrous form of CaSO4
Plaster of Paris and gypsum are the hydrated forms of calcium sulfate. They are the two well-known forms of hydrated calcium sulfate because they have many applications, especially in pharmaceutical and building industries. When calcium sulfate dihydrate (gypsum) is heated to 120-180 of calcium sulfate hemihydrate is produced that is also called plaster of Paris (POP). Physicians use POP to support broken bones during the healing process. POP can also be prepared by heating gypsum at 373 K. The related reaction is as follows:
CaSO4.2H2O (heated at 373 K) → CaSO4. 0.5 H2O + 1.5 H2O
Gypsum has the chemical name calcium sulfate dihydrate. Its chemical formula is CaSO4.2H2O. It is used as a protective and decorative finish for walls, ceilings and decorative pieces.
However, POP is a man-made material. When water is mixed with POP, it hardens and turns into gypsum. The related reaction is as follows:
CaSO4. 0.5 H2O + 1.5 H2O → CaSO4.2 H2O
Applications of calcium sulfate in industries
This mineral is used for many purposes some of which are as follows: it is used
- In construction works
- In decorative materials
- As mortar
- In casting, molds, and modeling
- In fertilizers and for soil aeration, as a soil amendment to improve soil efficiency and water absorption and to neutralize the corrosive effect of alkalinity
- As alabaster in sculpture
- As the main source of calcium in diets
- As a way of causing permanent hardness in water
- As an additive to turbid water, especially in ponds, for deposition of soil and clay particles without harming aquatic organisms
- In cooking
- As a component of Portland cement
- As a component of foot creams, shampoos and other products
- As a coloring agent for pharmaceuticals and cosmetic products
- Extensively in dentistry
- In growing mushrooms
- In removing carcinogenic contaminants such as lead and arsenic from contaminated water
Benefits of calcium sulfate as a dietary supplement
E516 (food grade calcium sulfate dihydrate) is used as a sequestrant in food and also as a buffer and thickening agent.
It also acts as a bleaching agent for bread rolls, and an artificial sweetener base for flour, tinned tomatoes, soy tofu, dried eggs, cheese products, and toothpaste.
Medical applications
This product can also be used in the pharmaceutical industry as filler in the manufacturing various types of pills, as a filler material and as a calcium-containing compound. It can be homogenized with the medications. In addition, pure calcium sulfate is used in many hygienic products and massage materials because it generates heat on body skin and opens skin interstices.
Calcium sulfate in agriculture
A substantial amount of produced calcium sulfate is used in the agriculture sector. It can be applied to soil as a fertilizer and improve plant growth.
Is calcium sulfate toxic?
Calcium sulfate is a non-toxic mineral and can be beneficial for humans, animals, plants, and the environment. It is nonflammable.







